Anderson, JC;
Grounds, H;
Reeves, S;
Taylor, PW;
(2014)
Improved synthesis of structural analogues of (-)-epicatechin gallate for modulation of staphylococcal β-lactam resistance.
Tetrahedron
, 70
(21)
3485 - 3490.
10.1016/j.tet.2014.03.052.

Preview |
PDF
Tetrahedron_2014_70_3485-90.pdf Download (426kB) |
![[thumbnail of JPG]](https://discovery-pp.ucl.ac.uk/1427787/2.hassmallThumbnailVersion/1-s2.0-S0040402014004001-fx1.jpg)  Preview |
Other (JPG)
1-s2.0-S0040402014004001-fx1.jpg Download (22kB) |
![[thumbnail of JPG Fig. 1. Naturally occurring galloyl catechins and non-natural targets.]](https://discovery-pp.ucl.ac.uk/1427787/3.hassmallThumbnailVersion/1-s2.0-S0040402014004001-gr1.jpg)  Preview |
Other (JPG Fig. 1. Naturally occurring galloyl catechins and non-natural targets.)
1-s2.0-S0040402014004001-gr1.jpg Download (26kB) |
![[thumbnail of JPG Scheme 1. Reagents and conditions (i) AD-mix-β®. (ii) HCl, MeOH. (iii) HC(OMe)3, PPTS cat. (iv) AcBr; K2CO3. (v) NaBH4.]](https://discovery-pp.ucl.ac.uk/1427787/4.hassmallThumbnailVersion/1-s2.0-S0040402014004001-sc1.jpg)  Preview |
Other (JPG Scheme 1. Reagents and conditions (i) AD-mix-β®. (ii) HCl, MeOH. (iii) HC(OMe)3, PPTS cat. (iv) AcBr; K2CO3. (v) NaBH4.)
1-s2.0-S0040402014004001-sc1.jpg Download (12kB) |
![[thumbnail of JPG Scheme 3. Reagents and conditions ]](https://discovery-pp.ucl.ac.uk/1427787/5.hassmallThumbnailVersion/1-s2.0-S0040402014004001-sc3.jpg)  Preview |
Other (JPG Scheme 3. Reagents and conditions )
1-s2.0-S0040402014004001-sc3.jpg Download (31kB) |
![[thumbnail of JPG Scheme 2. Reagents and conditions ]](https://discovery-pp.ucl.ac.uk/1427787/6.hassmallThumbnailVersion/1-s2.0-S0040402014004001-sc2.jpg) 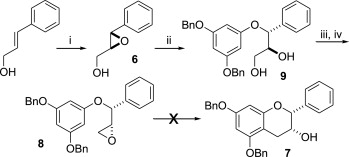 Preview |
Other (JPG Scheme 2. Reagents and conditions )
1-s2.0-S0040402014004001-sc2.jpg Download (18kB) |
![[thumbnail of JPG Scheme 4. Reagents and conditions]](https://discovery-pp.ucl.ac.uk/1427787/7.hassmallThumbnailVersion/1-s2.0-S0040402014004001-sc4.jpg) 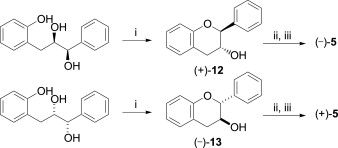 Preview |
Other (JPG Scheme 4. Reagents and conditions)
1-s2.0-S0040402014004001-sc4.jpg Download (16kB) |
Abstract
The high-yielding synthesis of enantiomerically pure epicatechin gallate analogues where the A and/or B-ring hydroxylation is reduced or altered has been achieved by optimising routes to the catechin stereochemistry. The B-ring analogues were synthesised by using an electrophilic ring closure onto an enantiomerically enriched epoxide as a key step. The A and B-ring hydroxyl-deleted analogues were synthesised through a Mitsunobu cyclisation. For the B-ring analogues, the anti- (catechin) stereochemistry was converted to the syn- (epicatechin) stereochemistry by a known oxidation/reduction protocol. Absolute stereochemistry was derived from either a Sharpless epoxidation or asymmetric dihydroxylation.
Archive Staff Only
 |
View Item |


